

Chinese General Practice ›› 2022, Vol. 25 ›› Issue (21): 2661-2669.DOI: 10.12114/j.issn.1007-9572.2022.0090
Special Issue: 骨健康最新文章合辑
• Evidence-based Medicine • Previous Articles Next Articles
Received:2022-01-25
Revised:2022-03-18
Published:2022-07-20
Online:2022-05-19
Contact:
Hui GENG
About author:
通讯作者:
耿惠
作者简介:基金资助:
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.chinagp.net/EN/10.12114/j.issn.1007-9572.2022.0090
| 步骤 | 检索词 |
|---|---|
| #1 | Multiple Myeloma OR Multiple Myelomas OR Myelomas,Multiple OR Myeloma,Multiple OR Myeloma,Plasma-Cell OR Myeloma,Plasma Cell OR Myelomas,Plasma-Cell OR Plasma-Cell Myeloma OR Plasma-Cell Myelomas OR Myelomatosis OR Myelomatoses OR Plasma Cell Myeloma,Plasma OR Cell Myelomas,Plasma OR Myelomas,Plasma Cell OR Plasma Cell Myelomas OR Kahler Disease OR Disease,Kahler OR Myeloma-Multiple OR Myeloma Multiple OR Myeloma-Multiples |
| #2 | randomized controlled trial OR randomized OR placebo |
| #3 | Daratumumab OR humax-CD38 OR Darzalex |
| #4 | Isatuximab OR Sarclisa OR SAR650984 OR SAR |
| #5 | (#1 AND #2) AND #3 |
| #6 | (#1 AND #2) AND #4 |
Figure 1 Strategy for searching randomized controlled trials of CD38 monoclonal antibodies treating relapsed and refractory multiple myeloma in Web of Science
| 步骤 | 检索词 |
|---|---|
| #1 | Multiple Myeloma OR Multiple Myelomas OR Myelomas,Multiple OR Myeloma,Multiple OR Myeloma,Plasma-Cell OR Myeloma,Plasma Cell OR Myelomas,Plasma-Cell OR Plasma-Cell Myeloma OR Plasma-Cell Myelomas OR Myelomatosis OR Myelomatoses OR Plasma Cell Myeloma,Plasma OR Cell Myelomas,Plasma OR Myelomas,Plasma Cell OR Plasma Cell Myelomas OR Kahler Disease OR Disease,Kahler OR Myeloma-Multiple OR Myeloma Multiple OR Myeloma-Multiples |
| #2 | randomized controlled trial OR randomized OR placebo |
| #3 | Daratumumab OR humax-CD38 OR Darzalex |
| #4 | Isatuximab OR Sarclisa OR SAR650984 OR SAR |
| #5 | (#1 AND #2) AND #3 |
| #6 | (#1 AND #2) AND #4 |
| 第一作者 | 发表年份(年) | 既往治疗次数(次) | 随访时间(月) | 纳入例数 | 年龄(岁) | 干预措施 | ORR(%) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 试验组 | 对照组 | 试验组 | 对照组 | 试验组 | 对照组 | |||||
| MARTIN[ | 2020 | 1~3 | 20.7 | 302 | 64(33,90) | IKd∶179 | Kd∶123 | 86.6 | 82.9 | |
| DIMOPOULOS[ | 2021 | ≥3 | — | 164 | 66(42,85) | 68(37,84) | Id∶55 | I∶109 | 43.6 | 23.9 |
| ATTAL[ | 2019 | ≥2 | 11.6 | 307 | 68(60,74) | 66(59,71) | IPd∶154 | Pd∶153 | 33.1 | 28.1 |
| DIMOPOULOS[ | 2020 | 1~3 | 17 | 466 | 64(57,70) | 65(59,71) | DKd∶312 | Kd∶154 | 84.3 | 74.7 |
| MATEOS[ | 2020 | ≥1 | 40 | 498 | 64(30,88) | 64(33,85) | DVd∶251 | Vd∶247 | 84.6 | 63.2 |
| BAHLIS[ | 2020 | ≥1 | 44.3 | 569 | 65(34,89) | 65(42,87) | DRd∶286 | Rd∶283 | 92.9 | 76.4 |
| DIMOPOULOS[ | 2021 | ≥1 | 16.9 | 304 | 67(42,86) | 68(35,90) | DPd∶151 | Pd∶153 | 68.9 | 46.4 |
| LU[ | 2021 | ≥1 | 8.2 | 211 | 61(28,79) | 61(43,82) | DVd∶141 | Vd∶70 | 82.5 | 58.6 |
Table 2 Basic characteristics of the included randomized controlled trials
| 第一作者 | 发表年份(年) | 既往治疗次数(次) | 随访时间(月) | 纳入例数 | 年龄(岁) | 干预措施 | ORR(%) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 试验组 | 对照组 | 试验组 | 对照组 | 试验组 | 对照组 | |||||
| MARTIN[ | 2020 | 1~3 | 20.7 | 302 | 64(33,90) | IKd∶179 | Kd∶123 | 86.6 | 82.9 | |
| DIMOPOULOS[ | 2021 | ≥3 | — | 164 | 66(42,85) | 68(37,84) | Id∶55 | I∶109 | 43.6 | 23.9 |
| ATTAL[ | 2019 | ≥2 | 11.6 | 307 | 68(60,74) | 66(59,71) | IPd∶154 | Pd∶153 | 33.1 | 28.1 |
| DIMOPOULOS[ | 2020 | 1~3 | 17 | 466 | 64(57,70) | 65(59,71) | DKd∶312 | Kd∶154 | 84.3 | 74.7 |
| MATEOS[ | 2020 | ≥1 | 40 | 498 | 64(30,88) | 64(33,85) | DVd∶251 | Vd∶247 | 84.6 | 63.2 |
| BAHLIS[ | 2020 | ≥1 | 44.3 | 569 | 65(34,89) | 65(42,87) | DRd∶286 | Rd∶283 | 92.9 | 76.4 |
| DIMOPOULOS[ | 2021 | ≥1 | 16.9 | 304 | 67(42,86) | 68(35,90) | DPd∶151 | Pd∶153 | 68.9 | 46.4 |
| LU[ | 2021 | ≥1 | 8.2 | 211 | 61(28,79) | 61(43,82) | DVd∶141 | Vd∶70 | 82.5 | 58.6 |
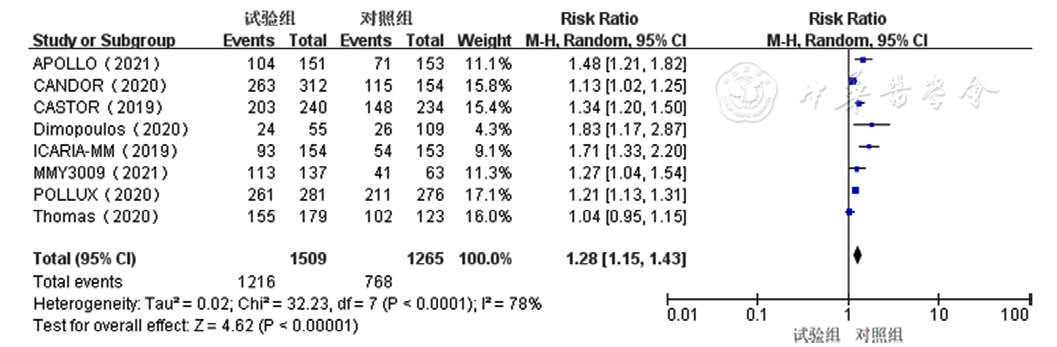
Figure 3 Forest plot of rates of overall response to treatment for relapsed and refractory multiple myeloma in the experimental group and control group
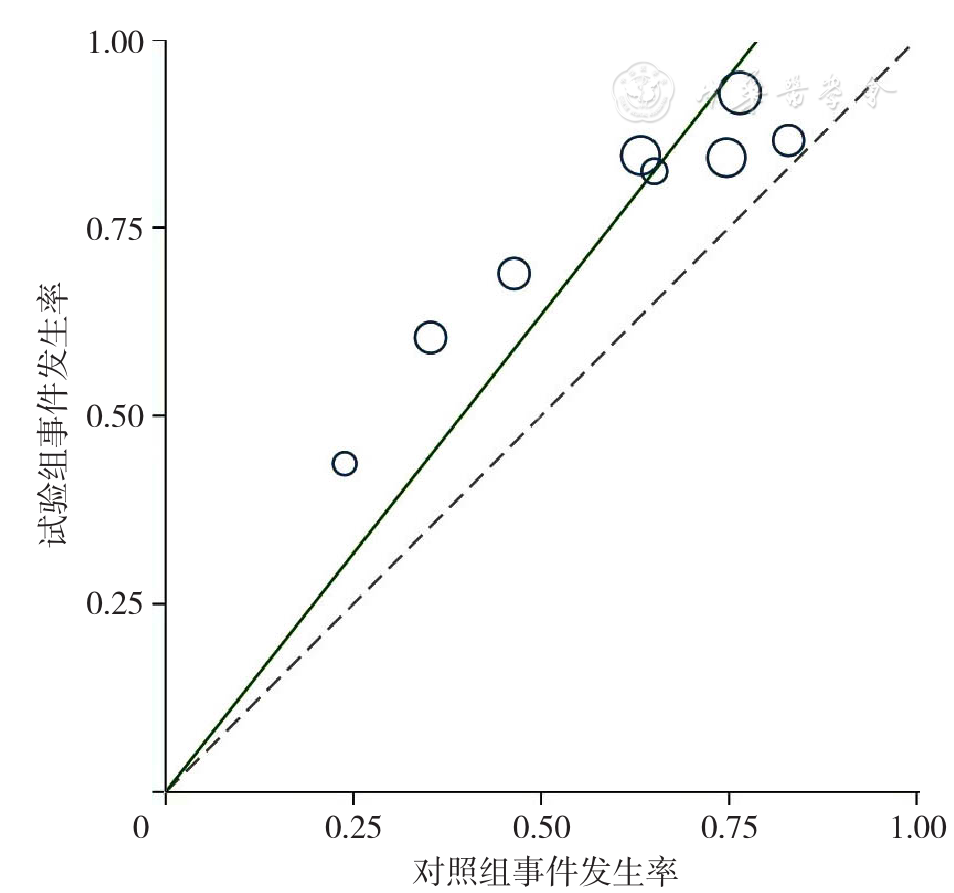
Figure 4 L'Abbe plotof rates of overall response to treatment for relapsed and refractory multiple myeloma in the experimental group and control group
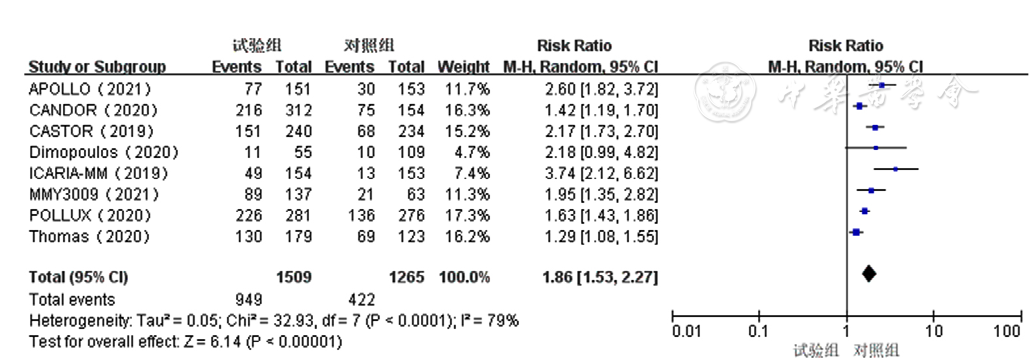
Figure 6 Forest plot assessing≥very good partial response to treatment for relapsed and refractory multiple myelomain the experimental group and control group
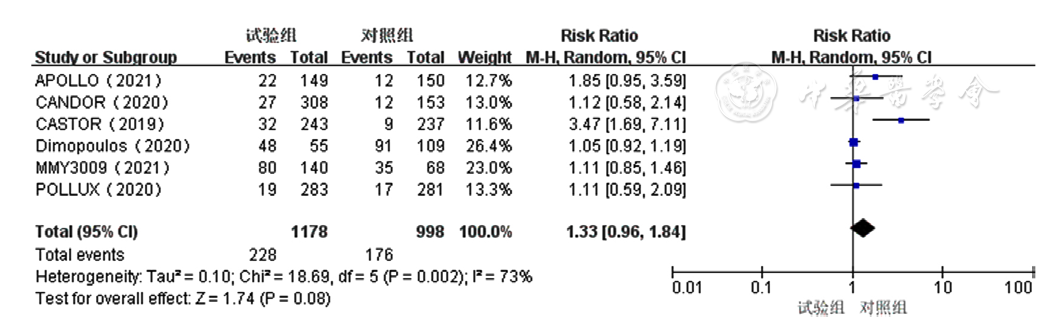
Figure 7 Forest plot assessing lymphocytopenia caused by treatment for relapsed and refractory multiple myeloma in the experimental group and control group
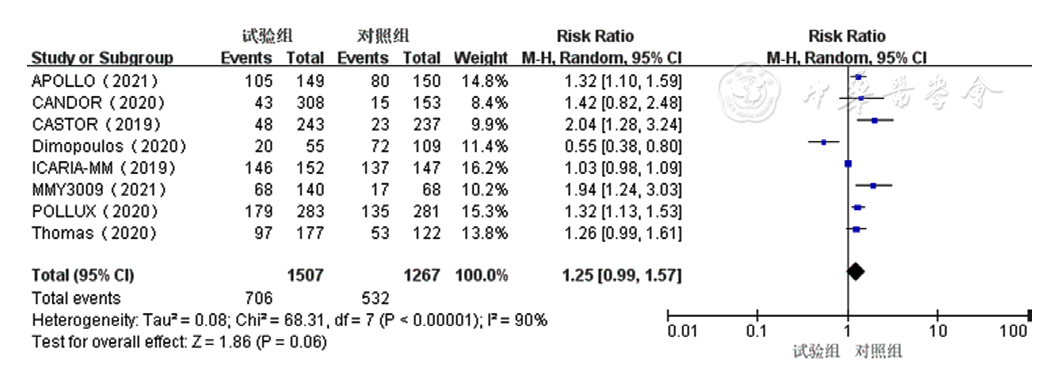
Figure 9 Forest plot assessing thrombocytopenia caused by treatment for relapsed and refractory multiple myeloma in the experimental group and control group
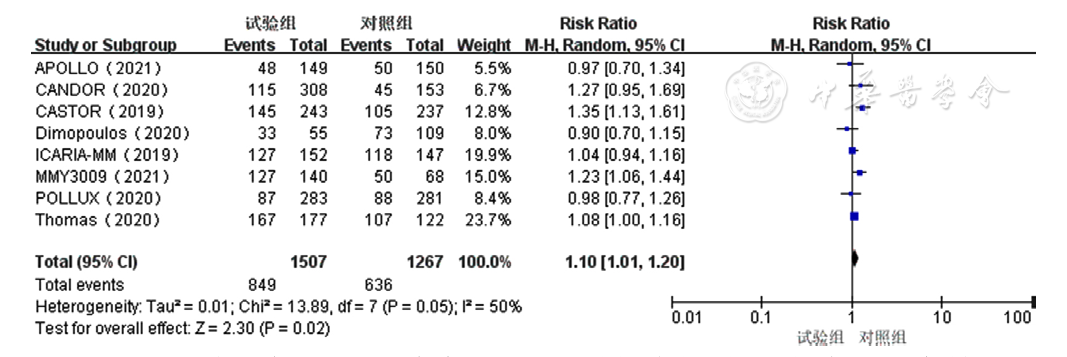
Figure 10 Forest plot assessing neutropenia caused by treatment for relapsed and refractory multiple myeloma in the experimental group and control group
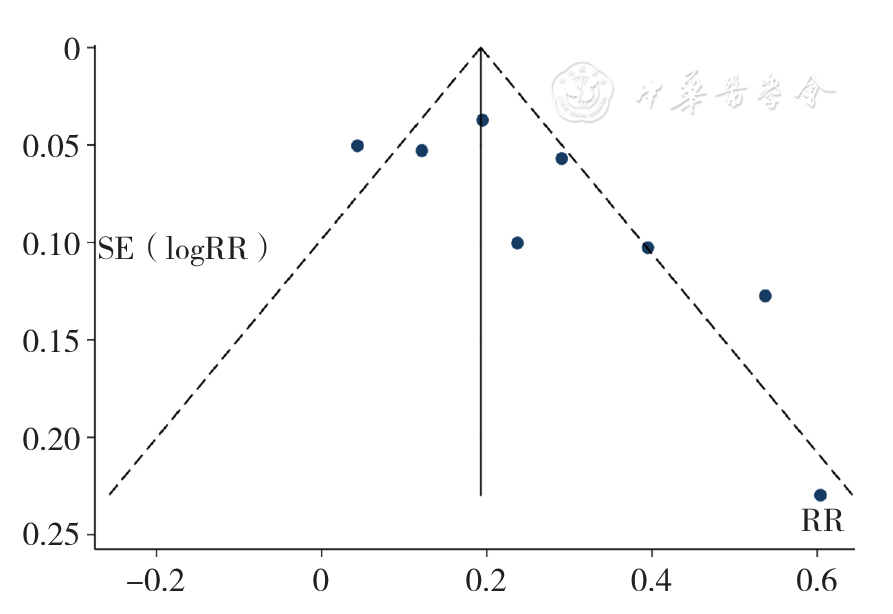
Figure 11 Funnel plot assessing publication bias of the rate of overall response to treatment for relapsed and refractory multiple myeloma between the experimental group and control group
| Std_Eff | 截距 | SE | t值 | P>|t| | 95%CI |
|---|---|---|---|---|---|
| slope | 0.035 | 0.081 | 0.43 | 0.679 | (-0.163,0.235) |
| bias | 2.807 | 1.305 | 2.15 | 0.075 | (-0.386,6.000) |
Table 4 Egger's test assessing potential publication bias
| Std_Eff | 截距 | SE | t值 | P>|t| | 95%CI |
|---|---|---|---|---|---|
| slope | 0.035 | 0.081 | 0.43 | 0.679 | (-0.163,0.235) |
| bias | 2.807 | 1.305 | 2.15 | 0.075 | (-0.386,6.000) |
| [1] |
|
| [2] |
《中国多发性骨髓瘤诊治指南(2020年修订)》发布[J].中华医学信息导报,2020,35(11):12.
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
王良哲,刘惠敏,侯健. 多发性骨髓瘤发病机制研究新进展[J]. 国际输血及血液学杂志,2007,30(4):319-322. DOI:10.3760/cma.j.issn.1673-419X.2007.04.008.
|
| [7] |
屈云,何俐,刘鸣. Cochrane系统评价的基本方法[J]. 中国临床康复,2003,7(4):532-533,536. DOI:10.3321/j.issn:1673-8225.2003.04.003.
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] | |
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [1] | ZHENG Boyue, FU Jiyi, WU Jiafei, WANG Jun, LI Hui. Study on the Efficacy and Safety of Carfilzomib in the Treatment of Multiple Myeloma [J]. Chinese General Practice, 2025, 28(30): 3806-3814. |
| [2] | CHEN Fei, WANG Jinying, YU Haibo, LI Xin, ZHANG Jiajia, SHEN Man, ZHAN Xiaokai, TANG Ran, FAN Sibin, ZHAO Fengyi, ZHANG Tianyu, HUANG Zhongxia. Significance of Elevated Urinary NGAL, TIM-1, VCAM-1 and Activin A in Patients with New Diagnosed Multiple Myeloma [J]. Chinese General Practice, 2025, 28(22): 2740-2749. |
| [3] | MA Panpan, WANG Sijing, YOU Na, DING Dafa, LU Yibing. Efficacy and Safety of Danuglipron and Orforglipron in the Treatment of Type 2 Diabetes Mellitus: a Meta-analysis [J]. Chinese General Practice, 2025, 28(21): 2679-2685. |
| [4] | ADILI Tuersun, CHENG Gang. Meta-analysis of the Efficacy and Safety of Finerenone in the Treatment of Type 2 Diabetic Nephropathy [J]. Chinese General Practice, 2025, 28(21): 2686-2691. |
| [5] | ZHU Shengjie, DIAO Huaqiong, HANG Xiaoyi, SUN Wenjun. Network Meta-analysis of Different Traditional Chinese Medicine Injections for the Treatment of Posterior Circulatory Ischemic Vertigo [J]. Chinese General Practice, 2025, 28(14): 1795-1808. |
| [6] | LIU Caiping, ZHANG Yanhua, TANG Jianpin, WANG Chengpeng, XUE Fengfeng, WANG Huijuan, LI Chuanwei, ZHANG Guangya, LI Huafang. Efficacy and Safety of Long-acting Risperidone Microspheres in the Maintenance Treatment of Schizophrenia [J]. Chinese General Practice, 2025, 28(13): 1622-1627. |
| [7] | SONG Fenfen, LI Shengmian. Real-World Study of Camrelizumab-based Regimen for Locally Advanced and Metastatic Esophageal Cancer [J]. Chinese General Practice, 2025, 28(07): 844-852. |
| [8] | MA Huping, REN Rong, HOU Mei, YUAN Aiyun. Clinical Observation of the New Antiepileptic Drug Perampanel in the Treatment of Refractory Epilepsy in Children Aged 0-18 Years [J]. Chinese General Practice, 2025, 28(02): 250-256. |
| [9] | KONG Yan, ZUO Yanli, LIU Jianghua, WU Huabei, CHEN Enran, WEI Siyu. Performance and Retention Intention Rates of Order-oriented Medical Students in China: a Meta-analysis [J]. Chinese General Practice, 2024, 27(28): 3485-3494. |
| [10] | YANG Can, LI Ning, LI Xuefei, ZHAO Li, XU Hao, SHI Qi, WANG Yongjun, LIANG Qianqian. Efficacy of Zang Bi Formula in Treating Arthritis and Its Pulmonary Complications in Rheumatoid Arthritis Interstitial Lung Disease Mice [J]. Chinese General Practice, 2024, 27(24): 3015-3022. |
| [11] | PANG Lan, LI Peifan, ZHU Xiaogang, YANG Zaihong, ZHENG Lei. Deep Transcranial Magnetic Stimulation Combined with Escitalopram Oxalate in the Treatment of Depression: a Randomized Controlled Trial [J]. Chinese General Practice, 2024, 27(17): 2098-2103. |
| [12] | ZHAO Hui, LI Wenhao, CHENG Gong, CHEN Liang, LIANG Chenyuan, WANG Yiyang, JIANG Hongying, JIANG Ruijia. Analysis of Adverse Reactions in the Assessment of Coronary Microvascular Disease by Adenosine Load and Adenosine Disodium Triphosphate Load [J]. Chinese General Practice, 2024, 27(17): 2109-2112. |
| [13] | ZHAO Yanan, HAN Shifan, LI Ying, ZHOU Liyuan, YANG Jie, WU Jiaxin, CHEN Ganggang. Efficacy of Virtual Reality Vestibular Rehabilitation Training in Patients with Sudden Deafness and Vertigo: a Randomized Controlled Trial [J]. Chinese General Practice, 2024, 27(14): 1672-1677. |
| [14] | NIU Jingyuan, CHEN Huisheng, YU Jiaxiang, CUI Yu. Ligustrazine Injection in the Treatment of Acute Ischemic Stroke: a Bayesian Network Meta-analysis [J]. Chinese General Practice, 2024, 27(14): 1761-1774. |
| [15] | ZHANG Yong, WANG Senli, HUANG Ronghua, XU Fengping, LIU Dan. Intervention Effect of Music Therapy on Patients with Alzheimers Disease: a Meta-analysis [J]. Chinese General Practice, 2024, 27(12): 1511-1518. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||